Atomic modEL LAB reflection
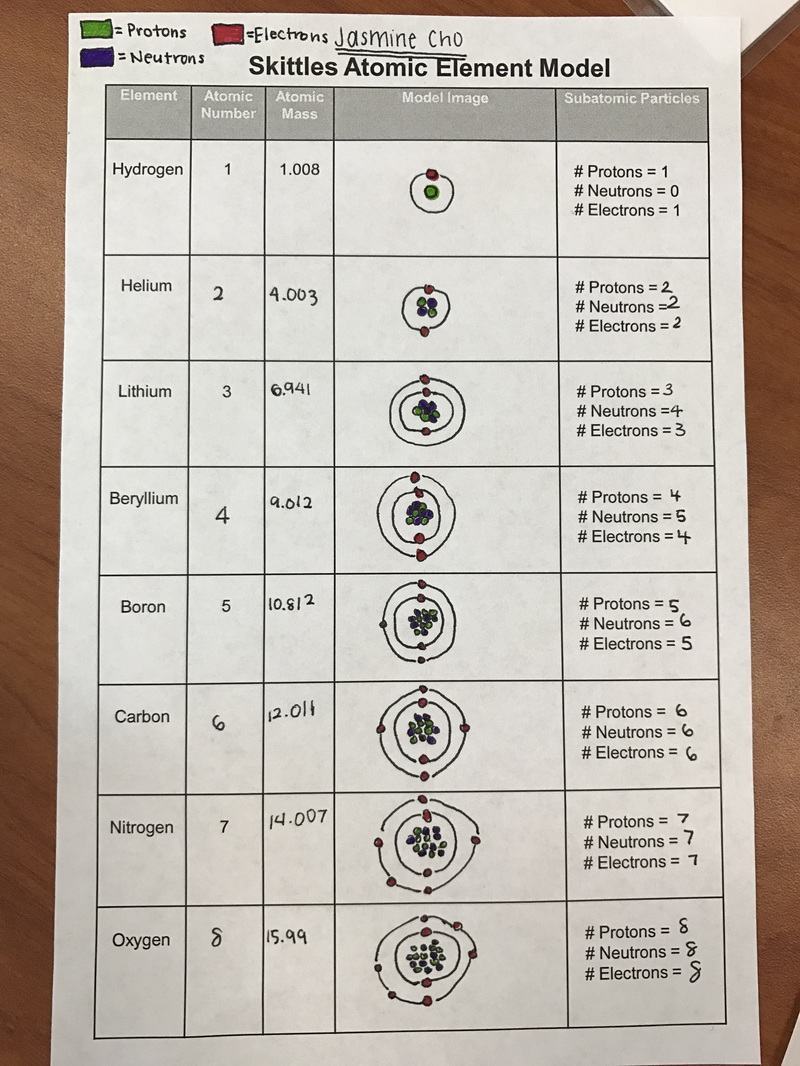
Today we conducted a lab where we created atomic models of the first eight elements on the periodic table- hydrogen, helium, lithium, beryllium, boron, carbon, nitrogen, and oxygen. Before doing this lab the basic information and knowledge I had about atoms was that there were three basic parts of an atom-protons = positive charge, neutrons = no charge, and electrons = negative charge. I also knew that there were two regions on an atom: the nucleus and the electron cloud. The nucleus was the center of the atom that contains the mass of the atom The electron cloud was the region that surrounds the nucleus that contains most of the space in the atom.
We got skittles and the green ones represented protons, the red ones represented electrons, and the purple skittles represented neutrons. We first found their atomic number and atomic mass that helped us determined the subatomic particle. Protons and electrons were equivalent to the atomic number and the number of neutrons was the atomic mass (rounded to the nearest number) subtracted with the atomic number. Then we modeled the element on the atomic structure handout and then transferred our model into a diagram on the skittles atomic element model sheet.
Throughout this lab, I learned that there was a connection within the subatomic particles. 1) protons= electrons= atomic number. 2) neutrons= atomic mass (rounded to nearest whole number) - atomic number. The one thing I noticed from our model to reality was that our model that we made wasn't accurate. Although the information of the number of electrons, protons, and neutrons were correct, the model wasn't to scale. The model not being to scale isn't that important when you are learning about an atom in particular, but would be important to know the size comparison between protons, electrons, and neutrons. The most important thing I learned with this lab was about the locations of the electrons in an atom. Electrons are arranged in energy levels or shells around the nucleus of an atom. The first shell had a maximum of two electrons. The second and third shell had a maximum of eight electrons.
We got skittles and the green ones represented protons, the red ones represented electrons, and the purple skittles represented neutrons. We first found their atomic number and atomic mass that helped us determined the subatomic particle. Protons and electrons were equivalent to the atomic number and the number of neutrons was the atomic mass (rounded to the nearest number) subtracted with the atomic number. Then we modeled the element on the atomic structure handout and then transferred our model into a diagram on the skittles atomic element model sheet.
Throughout this lab, I learned that there was a connection within the subatomic particles. 1) protons= electrons= atomic number. 2) neutrons= atomic mass (rounded to nearest whole number) - atomic number. The one thing I noticed from our model to reality was that our model that we made wasn't accurate. Although the information of the number of electrons, protons, and neutrons were correct, the model wasn't to scale. The model not being to scale isn't that important when you are learning about an atom in particular, but would be important to know the size comparison between protons, electrons, and neutrons. The most important thing I learned with this lab was about the locations of the electrons in an atom. Electrons are arranged in energy levels or shells around the nucleus of an atom. The first shell had a maximum of two electrons. The second and third shell had a maximum of eight electrons.